Veeva Site Connect
Simplify Information Sharing
with All of Your Sites
Reduce complexity for sites and improve clinical research.
Announced 2020 Status Early Customers 11-50
See how Site Connect transforms how sponsors and sites work together
Overview
Automate the flow of
information to and from sites
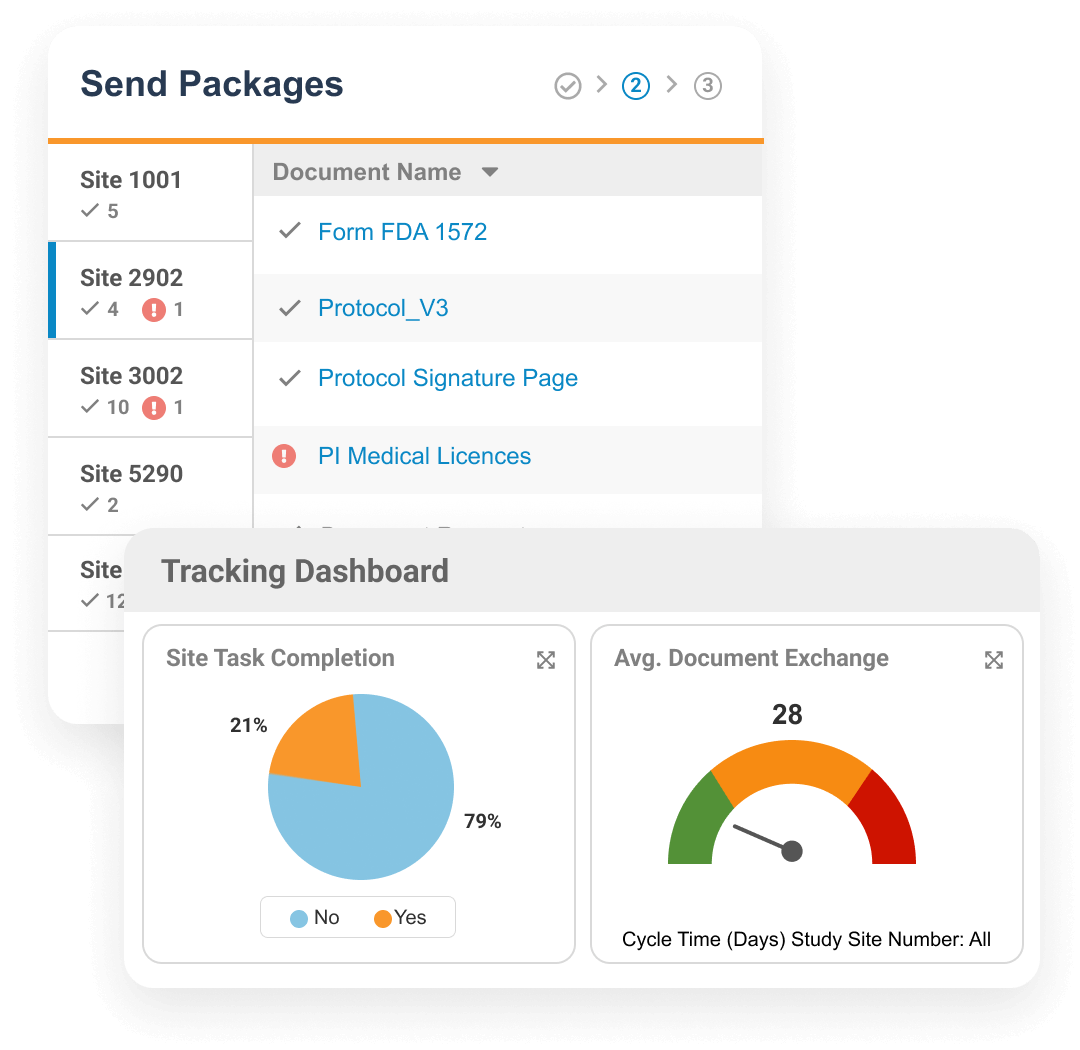
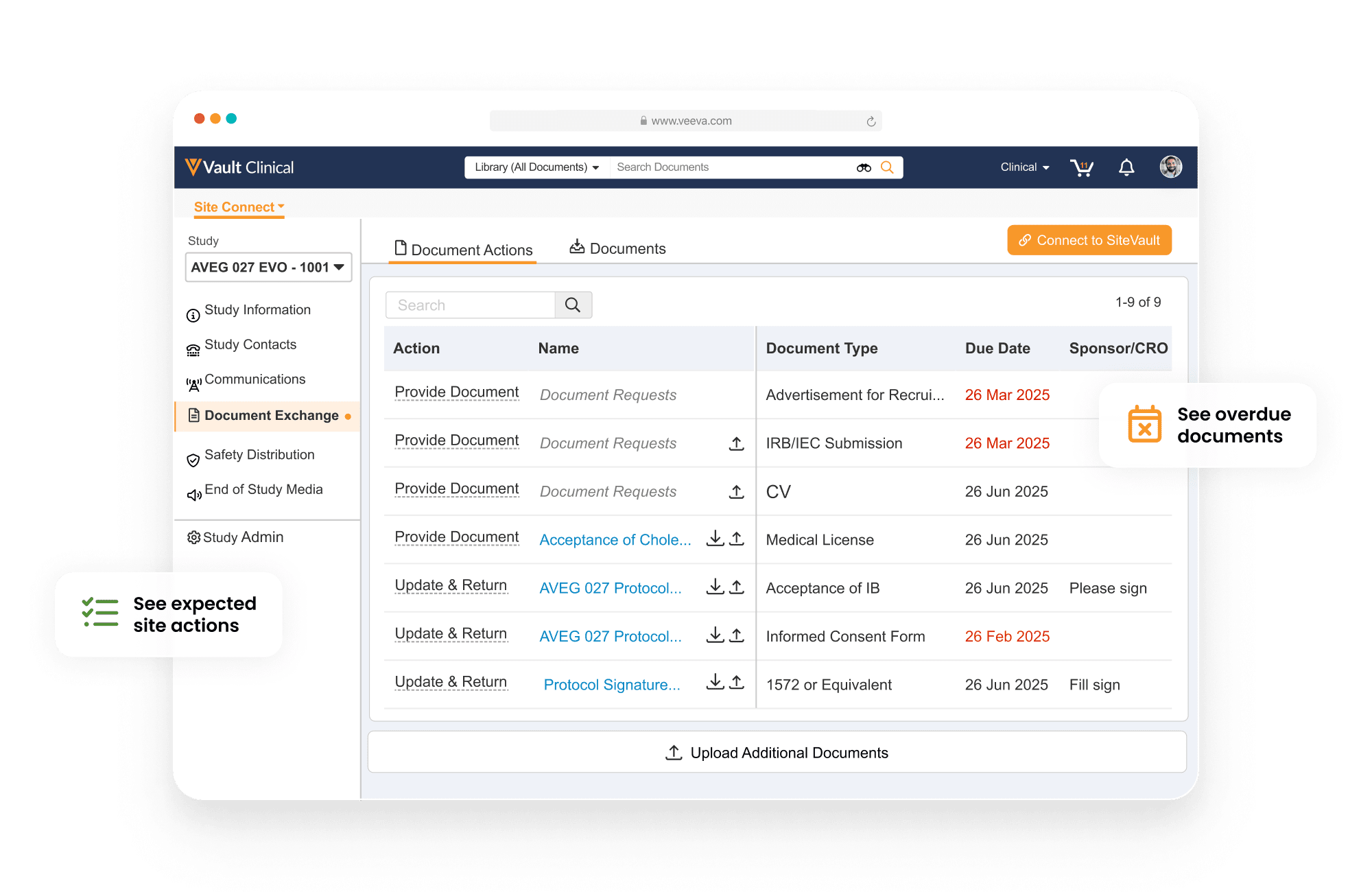
Veeva Site Connect allows sponsors and research sites to collaborate on a trial by automating the flow of information to and from sites during startup, execution, and closeout.
Information flow includes protocols, essential document packages, safety reports, and payment letters. Required media is sent on closeout, including completed CRFs. Information sent and received is automatically filed in eTMF.
Research sites manage tasks, documents, and data in Site Connect. Optionally, sites can connect their SiteVault for enhanced functionality.
Why Veeva Site Connect
Better collaboration and faster trials
Customer Success
Automating information sharing
for better collaboration