Veeva Vault CTMS
Manage Trials End-to-End
with a Modern CTMS
Proactively manage trials and accelerate toward
a more connected digital future.
Announced 2016 Status Very Mature Customers 100+
See how Vault CTMS streamlines trial management
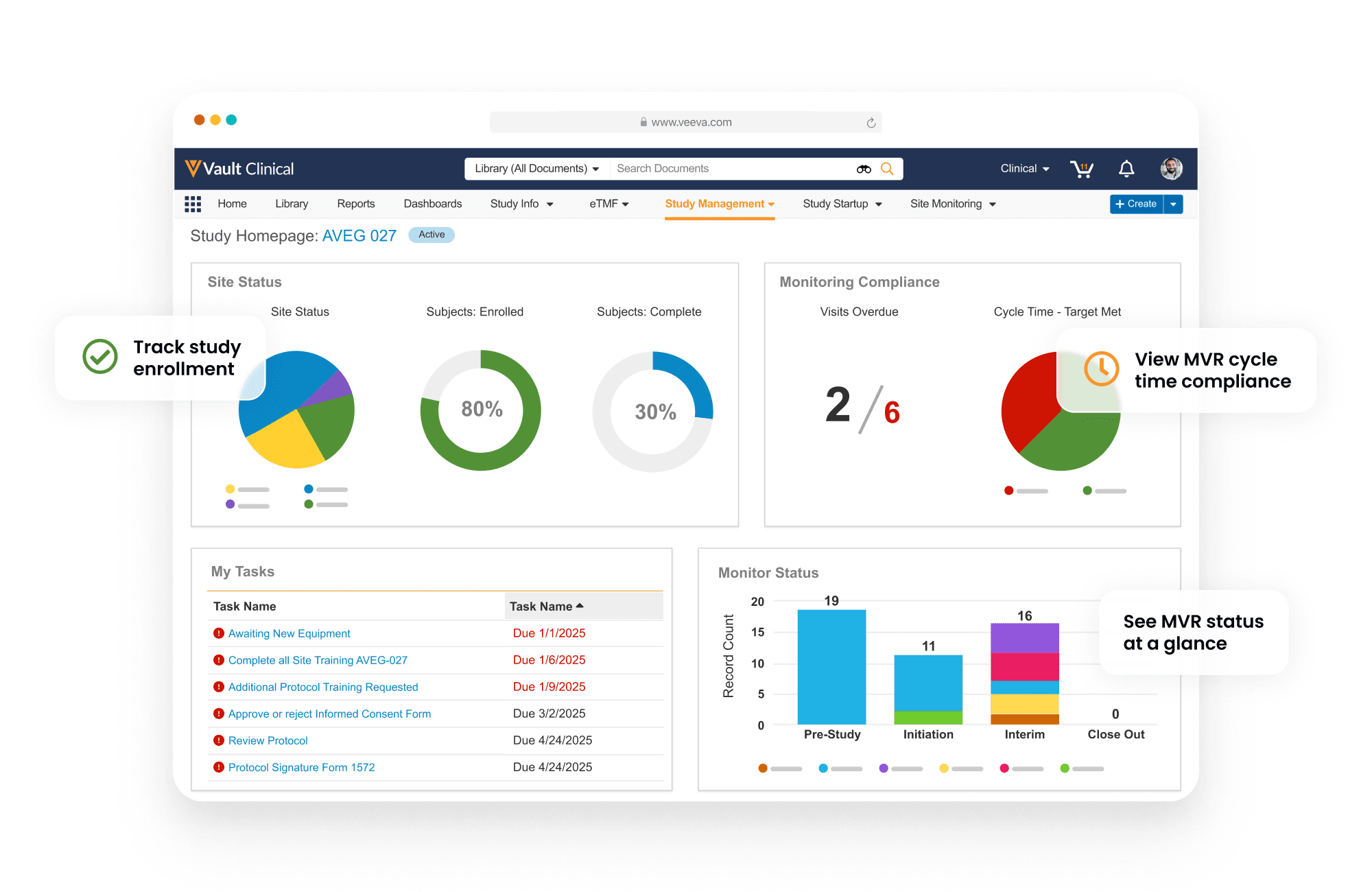
Overview
Easily manage insourced
and outsourced studies
with Vault CTMS
Enterprise trial management system
Veeva Vault CTMS provides end-to-end study management and monitoring capabilities for insourced and outsourced trials.
Full-featured to support trial needs
Dashboards and reports track key indicators, including enrollment and milestones, with drill-down to take action. Monitoring visit reports support automation and dynamic question branching. Trip reports are automatically filed within Vault eTMF. Issues and Protocol Deviations are logged as needed and routed through resolution workflows to ensure closure.
Connected with Vault EDC
Vault CTMS is connected with Vault EDC to support enrollment, monitoring, payments, and navigation to casebooks directly from within Vault CTMS. Investigator interactions synchronize with Veeva CRM for a 360-view.
Impact
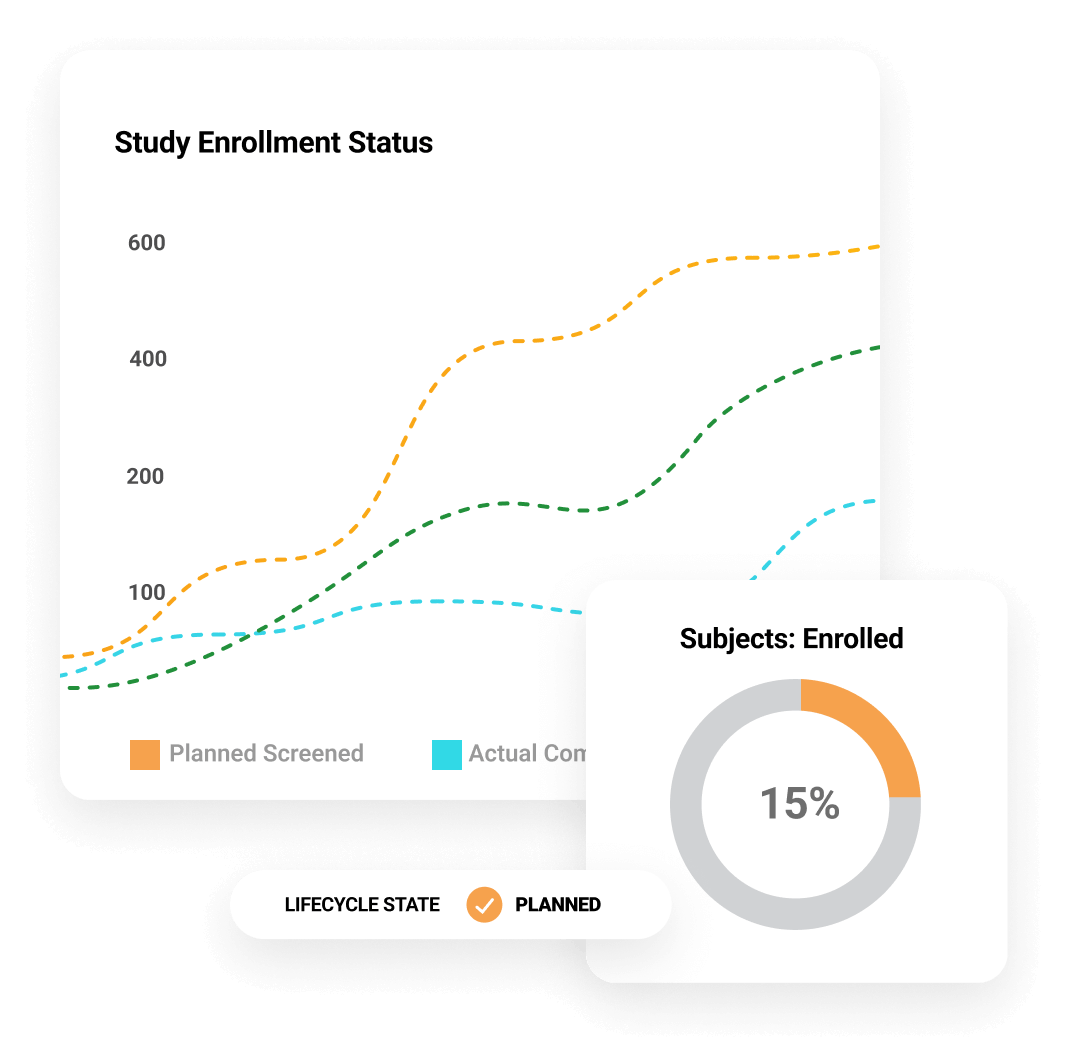
Efficiency at scale
30%
reduction in monitoring planning & execution
25%
improvement in issue management efficiency
465
days saved per 1,000 trip reports
Why Vault CTMS
Efficiency at scale
Customer Success
Streamlining trial management
for 200+ sponsors and CROs










Resources
Explore and Learn
Read case study
Top 20 pharma: CTMS transformation best practices

Watch demo
Effectively plan and conduct monitoring visits
Learn more
Achieving effective study oversight with a modern CTMS

Read white paper
Modernizing the hub of clinical operations

Watch demo
Identify, assess, remediate, and monitor risks

Hear from a top 20 pharma
Making the case to replace legacy CTMS
